Photos / Sounds
What
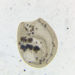
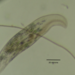
Genus CentropyxisObserver
brandoncorderDescription
Collected from plant detritus and surfaces in Razorback Lake, Wisconsin, 7/4/2024.
Photos / Sounds
What
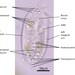
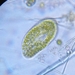
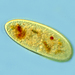
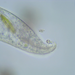
Genus LoxodesObserver
amayakanDescription
Video: https://youtu.be/Pr94XvqwciI
Photos are snapshots of the same individual featured in the video
Cell size: 204 µm in length
Site of sample collection: A wetland (rich in Spirogyra) near the pond, Takamori Ryokuchi (a freshwater habitat), Sendai, Japan
Date of sample collection: July 2nd 2022
Weather: Sunny
Water temp.: 28.6°C
pH 7.2
Date of observation: July 2nd 2022
Bright field observation using a Wraymer microscope (model BX-3500TL, Osaka, Japan) equipped with a Floyd-2 HDMI ethernet digital camera (Wraymer, Osaka, Japan). The accuracy of the scale bar was confirmed by using a stage micrometer glass slide (1 div. = 10 µm; Wraymer, Osaka, Japan) at each magnification.
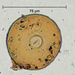
What
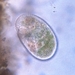
Genus ArcellaObserver
brandoncorderDescription
Collected from surface water in mucky edge of Minocqua Reservoir, 7/2/2024.
Photos / Sounds
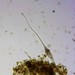
What
Genus LacrymariaObserver
brandoncorderDescription
Collected from surface water in mucky edge of Minocqua Reservoir, 7/2/2024.
Photos / Sounds
Observer
peptolabDescription
Pseudotrachelocerca trepida (Kahl, 1928) Song, 1990 from the sulfidic superficial intertidal benthos of marine estuary Acabonac Harbor. Imaged in Nomarski DIC on Olympus BH2S under SPlanap 40 0.95 and SPlan 100 1.25 oil immersion objectives plus variable phone camera cropping on Samsung Galxy S9+.
The cells mostly measure 140-180 um in length when fully extended showing a spindle shape but varying in that they show marked contractility of the neck region whereupon they assume a more club-shaped contour measuring 80 um. I found occasional otherwise identical individuals that exceeded 200 um (see final 7 pictures for the largest individual). Individuals are variably dorso-ventrally flattened but mostly the cells are more rounded in cross-section. The pellicle is grooved and textured with pits as first depicted by Kahl 1928 (Trachelocerca trepida) (1) and later described as grooved by Petz et al 1995 (2) and depicted in live photos of Hu, Lin and Song 2019 (3), though Song 1990 (4) in his redescription describes a smooth pellicle. The macronucleus is elongate and variably S-shaped or U-shaped as shown by all observers. Extrusomes are visible with difficulty seen primarily in the anterior 1/3 of the cell. Nematodesmata are quite difficult to visualize. The dorsal brush (or adoral organelle of Petz et al 1995), whose irregular arrangement rather than in rows, was very difficult to visualize in vivo as noted by Song 1990. These features point to a diagnosis of Pseudotrachelocerca trepida (Kahl, 1928) Song, 1990. I shall present all available descriptions and end with a discussion of the current thinking with regards to placement of the genus.
Kahl's initial description of Trachelocerca trepida: "Trachelocerca trepida KAHL, 1928 Size stretched 140 um, contracted (ovoid) 70 um; Body flattened (2:1). Neck tapered about half the width, contractile to the anterior end. core sausage-shaped, medium wide (20-24 heights). Neck to front end contractile. Special decoration row (dorsal brush) of the neck is not very noticeable. Near the left edge of a broad side of the neck a hyaline stripe on which a very dense series of long, backward curved cilia pulls, which can be seen towards the back of the body until about the middle which is (not easily recognizable). Cilia long and quite dense and inserted in pits. Trichocysts difficult to visualize in the throat (or at least very indistinct). Distributed in stronger 0Idesloe salt sites, oligosapropel, never numerous; occasionally in Sylt seawater ditch (160 um); but not examined in more detail here" (1).
Song's 1990 redescription: "New diagnosis. In vivo about 80-160 x 30 um; neck region somewhat contractile; body bottle-shaped, sometimes slightly flattened dorsoventrally. Macronucleus U or S-shaped; single contractile vacuole posteriorly; Extrusomes (toxicysts) genus-specific, mostly around the cytostome; 28 meridional somatic kineties on average. Redescription. Body shape in vivo moderately variable, anterior third neck-like narrowed, can be slightly curved in all directions. Anterior end more or less coned but not truncated as depicted by Kahl, posteriorly rounded, sometimes bag-like. Oral opening oval to long elliptic, lies polar on small, indistinct oral bulge, hardly recognizable in vivo. Nematodesmata not recognizable in vivo. Cilia ca. 5-8 um long, which at the oral end (circumoral ciliation) increase to ca. 10 um and, like that of Phialina, form a tuft with 5 girdles of cilia and beat anteriorly when feeding. Cilia of dorsal brush about 4-5 um, very difficult to recognize in living cells. Pellicle thin and smooth, not indented by ciliary rows as described by Kahl; beneath pellicle no special granules recognisable. Extrusomes occur both around cytostome and within cytoplasm,short rod-or spindle-shaped, often heavily stained with protargol. Cytoplasm very hyaline, colorless, usually densely filled with numerous dark grey, ca. 2 um particles. Macronucleus usually horseshoe-shaped, centrally located, with numerous nucleoli; micronucleus not found, presumably too near to macronucleus. Movement moderately rapid, usually swimming ahead in straight line while rotating around longitudinal body axis. Feeds possibly mainly on bacteria both in nature and in culture.
Somatic kineties course longitudinally, composed of monokinetids; distance between basal bodies smaller anteriorly than posteriorly. At base of oral bulge a circumoral kinety composed of dikinetids is present; the number of the basal body pairs of this kinety is smaller than that of somatic kinetics. After protargol impregnation the rhabdos is composed of, very probably, only one component: the nematodesmal bundles which originate from the anterior kinetosomes of the oral dikinetids. Dorsal brush very characteristic: made up of many basal body pairs which are "piled up" irregularly, unlike those in other haptorid families, where it forms regular rows. Brush extends posteriorly to about one sixth of the body length and, in addition, is connected with only one somatic kinety" (4).
Petz, Song and Wilbert described a population from icy waters of the Weddell Sea: " seudotrachelocerca trepida (KAHL, 1928a) SONG, 1990: In vivo 90-110 x 30-37 um. Body elongate club- to spindle-shaped, anteriorly narrowed, widest at or behind mid-body, frontal end straight, posteriorly rounded; contractile. Cross section elliptical to circular. Cytostome apical, not protruding, very likely circular. Pellicle thin, slightly grooved by kineties. Cortical granules colourless. Single macronucleus sausage- to U-shaped, located in mid-body, contains spherical (up to 3 urn across) and, rarely, elongate nucleoli. Once 3 micronuclei, globular, in indentation of macronucleus, usually not impregnated with protargol. Single contractile vacuole terminally. Cortical granules in rows along somatic kineties. Cytoplasm colourless, contains many greenish shining globules (1.5-4 um across) and food vacuoles with unidentified contents; specimens usually appear rather dark. Movement slow. Somatic kineties meridional, extending over entire length of body, especially anterior portions composed of very densely spaced monokinetids, cilia about 10 um long; parasomal sac adjacent to each basal body in silver nitrate slides (cf. FOISSNER 1983). Argentophilic structure (fibres?) immediately beneath each kinety. Somatic kineties terminate anteriorly with single dikinetid forming circumoral kinety. Adoral organelle (FOISSNER et al. 1994; formerly brosse) parallel to somatic kineties, i.e. aklitolophic (HILLER & BARDELE 1988), 28-51% of body length (x = 41%, n = 7), composed of numerous irregularly arranged dikinetids, distally indistinctly 2- to 3-rowed, proximally single-rowed, continued posteriad in 1 row of monokinetids (short somatic kinety); cilia about 10 um long. Extrusomes spindle-shaped, usually slightly bent, about 0.5 um thick, sometimes with thread-like process, scattered in body, not found surrounding cytostome (not impregnated?), once, however, observed outside cytostome (preparation artifact?); in protargol slides, extruded extrusomes very fine, ca. 20 um long. Nematodesmata fine, about 23 um long. Occurrence and ecology: Infrequently found in the endopagial of pancake and multiyear sea ice of the Weddell Sea, between latitude 69° 07-70° 30' S and longitude 07° 19'-12° 08' W. Occurs together with diatoms, autotrophic and heterotrophic flagellates and ciliates "(4).
Discussion from Petz et al 1995: " Comparison with related species and systematic considerations: The irregularly arranged adoral organelle is a main character of the monotypic genus Pseudotrachelocerca (SONG 1990). The Antarctic specimens are quite similar to P. trepida (AGAMALIEV 1983; BORROR 1965; KAHL 1928a; SONG 1990; WILBERT & KAHAN 1981). They differ slightly in the possession of cortical granules (perhaps a variable character) and a longer adoral organelle (1/3 1/2 vs. 1/6-1/4 of body length; AGAMALIEV 1983; KAHL 1928a, 1932; SONG 1990; WILBERT & KAHAN 1981). The latter depends, however, on the contraction of the specimens. The population studied by BORROR (1965) also has a long adoral organelle, thus we identify the present organisms with P. trepida. The adoral organelle arrangement and the meridional somatic kineties suggest that Pseudotrachelocerca is related to holophryids (FOISSNER et al. 1994; HILLER & BARDELE 1988). This genus is thus transferred from the Spathidiida to the Prostomatida. The family Holophryidae PERTY, 1852 is, however, distinguished from Pseudotrachelocercidae SONG, 1990 in the 3-rowed adoral organelle (FOISSNER et al. 1994; HILLER & BARDELE 1988). [Due to the rather complicated nomenclature and taxonomy in prostomatids, Prorodontidae of HILLER & BARDELE (1988) are Holophryidae (FOISSNER et al. 1994)]" (4).
Finally, Huang et al 2019 analyzed 14 small subunit ribosomal RNA gene sequences, 13 large subunit ribosomal RNA gene sequences and 13 ITS1- 5.8S- ITS2 gene sequences of various haptorians including the first molecular data from Pseudotrachelocerca Song, 1990: "The historical assignment of Pseudotrachelocerca trepida to the class Litostomatea is rejected by our analyses; rather, it clusters with Cyclotrichium cyclokaryon (a planktonic Mesodiunium-like organism) with high or full support and shows close relationships to classes Colpodea, Prostomatea and Plagiopylea, in all our single- gene and concentrated trees. Hitherto, Pseudotrachelocerca trepida has been assigned to the monogeneric family Pseudotrachelocercidae. Pseudotrachelocercica is similar to other litostomateans in having a dorsal brush (DB), apical extrusomes and a mesh- like silver line system (Song, 1990). Among these characters, the DB is an important feature for the classification of litostomateans, and a three- rowed DB is considered as an apomorphy of the last common ancestor (Vd’ačný et al., 2011). However, Pseudotrachelocerca is divergent in that its DB is composed of irregularly arranged kinetids and is connected to only one somatic kinety (vs. to several somatic kineties in most other litostomateans; Song, 1990). Both the divergent phylogenetic position and the heterogeneities in the DB appearance imply that the DB structure of Pseudotrachelocerca might be a homoplasy of the typical litostomatean dorsal brush. In addition, the topology testing results strongly reject the grouping of Pseudotrachelocerca to either Haptoria or Litostomatea (AU test, ρ< .05; Table 2). Accordingly, we suggest an exclusion of Pseudotrachelocercidae from the class Litostomatea" (5).
"Although Pseudotrachelocera grouped with Cyclotrichium with moderate- to- high support in all the phylogenetic analyses, there is no distinct morphological evidence to support this phylogenetic affiliation. Morphologically, Pseudotrachelocerca is similar to Cyclotrichium in possessing both oralized somatic monokinetids and circumoral dikinetids, characters that are also commonly present in some typical haptorians, for example Chaenea (Kwon, Vďačný, Shazib, & Shin, 2014; Xu, Song, & Hu, 2005). Other genera that cluster with Pseudotrachelocerca and Cyclotrichium and which are systematically ambiguous include Askenasia, Paraspathidium and the prostomatean genus Plagiocampa. Previous studies have placed these taxa in the class Plagiopylea, which was not well supported by our results (Gao, Warren, et al., 2016; Zhang, Yi, Song, Al- Rasheid, & Warren, 2010; Zhang et al., 2012). Relationships between these genera remain unresolved pending enrichment of taxa and genes. Based on the current knowledge, we propose that Pseudotrachelocercidae should be regarded as incertae sedis within the Prostomeatea + Plagiopylea + Colpodea complex" (5). Finally, it is interesting that in this study, Pseudotrachelocerca clusters with Paraspathidium, another ciliate which I also identified in this sample of saprobic estuarine benthic saltwater biofilm.
- Kahl A. (1930): Urtiere oder Protozoa. I. Wimpertiere oder Ciliata (Infusoria). I. Allgemeiner Teil und Prostornata. Tierwelt Dtl., 18, 1-180.
- Taxonomy and ecology of the ciliate fauna (Protozoa, Ciliophora) in the endopagial and pelagial of the Weddell Sea, Antarctica Wolfgang PETZ, Weibo SONG & Norbert WILBERT. Stapfia 40, 1995 pp 15-17.
- Ciliate Atlas: Species Found in the South China Sea. Xiaozhong Hu • Xiaofeng Lin • Weibo Song Editors. Science Press Beijing, Springer. 2019. ISBN 978-981-13-5900-2 College of Life Science South China Normal University Guangzhou, Guangdong, China ISBN 978-981-13-5901-9 (eBook) https://doi.org/10.1007/978-981-13-5901-9, pp 84-5.
- Infraciliature and Silverline System of the Ciliate Pseudotrachelocerca trepida (Kahl, 1928), nov. gen., nov comb., and Establishment of a New Family, the Pseudotrachelocercidae nov fam. (Ciliophora, Haptoria). Weibo Song. Europ.J.Protistol. 26, 160-166 (1990)
- Further insights into the highly derived haptorids (Ciliophora, Litostomatea): Phylogeny based on multigene data. Jie B. Huang, Tengteng Zhang, Qianqian Zhang,Yuan Li, Alan Warren, Hongbo Pan, Ying Yan. Zoologica Scripta. 2018; 47:231–242.
Photos / Sounds
What
Genus SpirostomumObserver
manuel_helsinkiDescription
Sample of water from a ditch.
Photos / Sounds
What
Genus LoxodesObserver
manuel_helsinkiDescription
Sample of water from a ditch.
Photos / Sounds
What
Hypotrichs (Subclass Hypotrichia)Observer
manuel_helsinkiDescription
From a river sample.
Photos / Sounds
What
Genus FrontoniaObserver
manuel_helsinkiDescription
Water sample from a small ditch.
Stats
- 7819